Research / Registries
Cloud-based, multi-site clinical trial administration and patient access
Connecting the dots
between patients and research
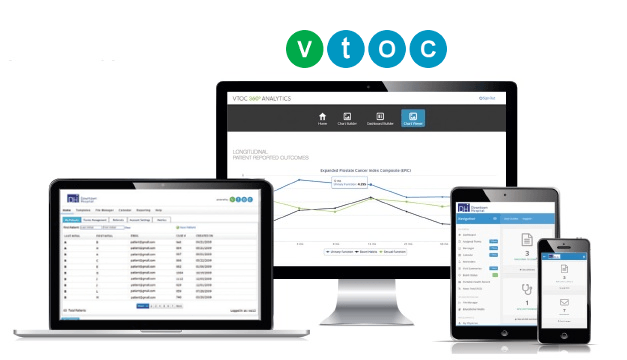
Visiontree for research & registries is a 21 CFR Part 11 compliant cloud-based, interoperable application for multi-site clinical trial management and patient access.
The Visiontree application provides a library of over 400 electronic patient and clinician reported outcomes assessments for ePRO and eCOA use. Visiontree allows you to manage decentralized clinical trials to bridge the gap between clinical research and clinical care.
Proven to deliver a high level of patient compliance, accuracy and validation, Visiontree provides data collection and curation with EHR interoperablity in semantic context of the patient health record for optimal multi-center research & registry management and reporting.
Research e-Forms

Clinical Trials Builder
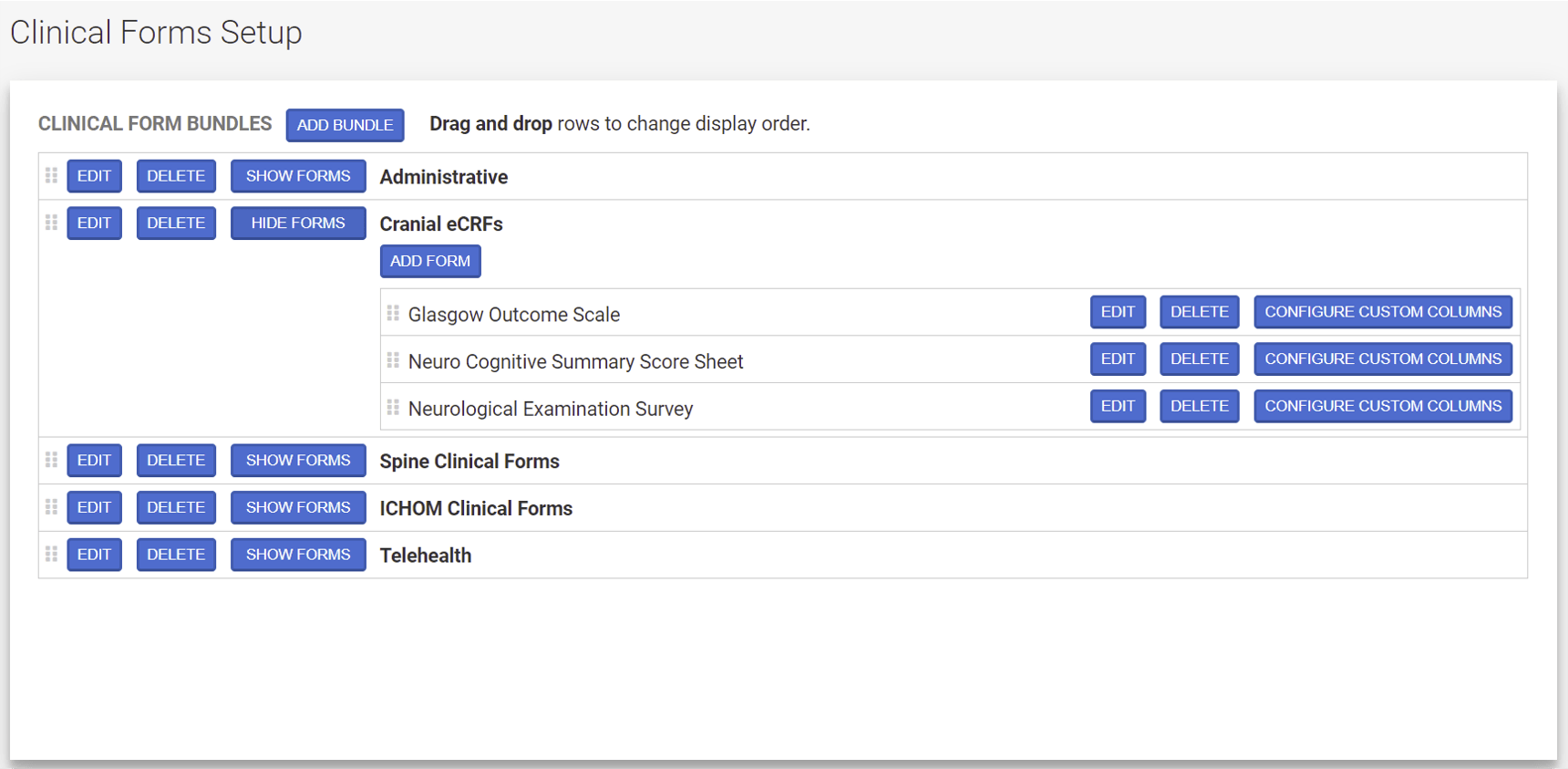
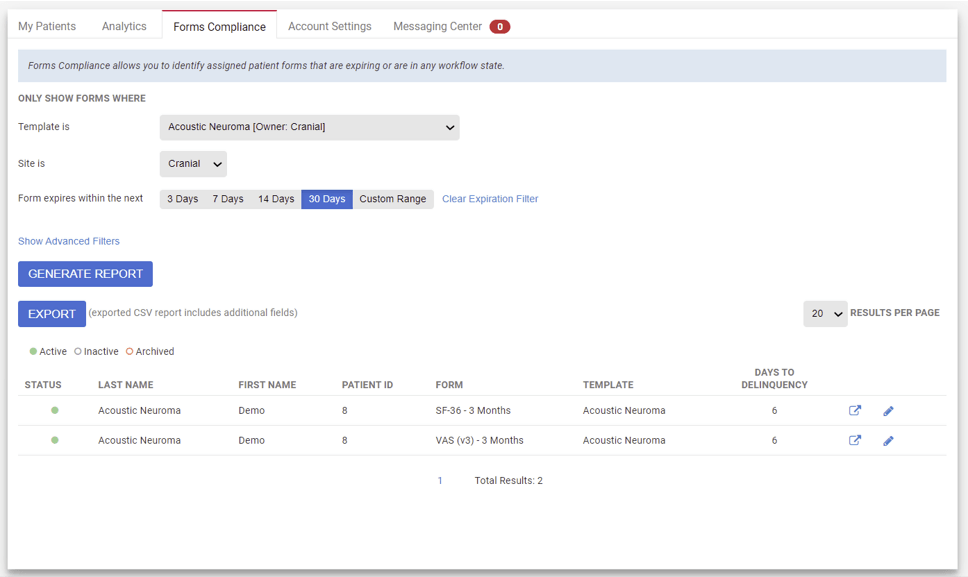
Compliance Manager
Outcomes Packages by Specialty
Visiontree provides standards-based ePRO, eCOA and customized solutions in these, and other, core domains.
- Oncology
- Cardiology &
Heart Surgery - Neurological &
Spine Surgery - Orthopaedic
Surgery - Behavioral
Health - Research &
Registries
Patient & Clinical Outcomes Data Collection & Reporting
Nimble and interoperable. The Visiontree Optimal Care™ (VTOC) cloud-based system allows providers to deliver improved quality and efficiency for patient care.
Efficient
- Save time with cloud-based, paperless workflow for all clinical research and patient reported outcomes forms
- Form compliance alerts for patients and research teams
- Access to an extensive library (1,000+) of electronic, validated electronic patient reported outcomes assessments (ePRO)
- Template and Timepoint Manager to build and manage your own research protocols
Accurate
- Electronic patient reported outcomes data collection and reporting at specific timepoints
- Research team and patient reminders by timepoint for high completion rates
- Proven to increase patient reported outcomes data collection rate by 40%+ over paper
- Template Manager allows for patients on multiple protocols, including separate treatment arms
Interoperable
- Integration with legacy EHR and research systems including Medidata Rave®
- Import Hub for data mapping of historical data and scheduled exports from EHR and disparate databases
- Connectivity with all data interchange standards (HL7, Web services)
- Multi-institutional participation with site-level access and Admin-level Aggregate data reporting
Interested?
For more information
